Equipment Monitoring: Cold Storage Monitoring
Cold Storage Temperature Monitoring
Internet of Things (IoT) technology is changing the way scientists collect and utilize data. By monitoring instrument and equipment status as well as the laboratory environment, scientists are able to better understand physical variables in the lab that may affect their research.
When considering a laboratory monitoring system, there are a number of variables to consider.
- MONITORING SHOULD BE RELIABLE AND CONVENIENT Critical to the success of any lab monitoring
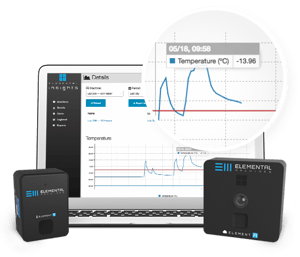 system is its usability. For any monitoring system to be remotely useful, data must be easy to access and readily available 24/7 to teams both on-site and to those working remotely. Preference should be given to products with intuitive designs and shared web-based dashboards that enable users to track the performance of critical instruments and equipment, to understand usage patterns to optimize operations, and to quickly identify potential issues that may arise. For many labs, change is inevitable, and flexibility is the key to success. Having the capacity to adapt quickly and easily to accommodate future projects is a necessary consideration for most lab managers. For this reason, equipment monitoring solutions should be scalable both user-wise and equipment-wise. Wireless and battery operated monitoring devices offer the easiest installation solution for laboratory monitoring, and when equipped with low-battery warnings and cellular backups they offer 24/7 operation with full confidence that data will always be available.
system is its usability. For any monitoring system to be remotely useful, data must be easy to access and readily available 24/7 to teams both on-site and to those working remotely. Preference should be given to products with intuitive designs and shared web-based dashboards that enable users to track the performance of critical instruments and equipment, to understand usage patterns to optimize operations, and to quickly identify potential issues that may arise. For many labs, change is inevitable, and flexibility is the key to success. Having the capacity to adapt quickly and easily to accommodate future projects is a necessary consideration for most lab managers. For this reason, equipment monitoring solutions should be scalable both user-wise and equipment-wise. Wireless and battery operated monitoring devices offer the easiest installation solution for laboratory monitoring, and when equipped with low-battery warnings and cellular backups they offer 24/7 operation with full confidence that data will always be available.
Laboratory plans and layouts change frequently. Cold storage temperature monitoring systems that require 120 VAC power and cabling for communications complicate moves and restrict layouts. Wireless systems like the Element-T from Elemental Machines simplify moving and layout as there are no cables to reroute, saving you time and money.
- COMPLIANCE IS CRITICAL Regardless of which quality system a lab is required to follow, be it GLP, GMP,
 ISO or CFR QSR, adopting a laboratory monitoring system will facilitate meeting the requirements for detailed, thorough record-keeping. Capturing time-stamped electronic data of test parameters will increase the efficiency of the lab while eliminating the likelihood of human error. Beyond compliance, captured data can be further utilized to drive process changes and optimize laboratory operations. For labs that fall under the jurisdiction of FDA compliance, 21 CFR Part 11 specifies requirements for electronic data collection is trustworthy, reliable, and equivalent to paper records. FDA regulated labs require solutions that offer complete audit trails. Events such as user logins and changes to key parameters that may affect process outcomes must be tracked. The lab monitoring system must also include user management and data security features such that only authorized users may access the system, that data measurements are non-editable, and that event details are time-stamped to ensure data integrity.
ISO or CFR QSR, adopting a laboratory monitoring system will facilitate meeting the requirements for detailed, thorough record-keeping. Capturing time-stamped electronic data of test parameters will increase the efficiency of the lab while eliminating the likelihood of human error. Beyond compliance, captured data can be further utilized to drive process changes and optimize laboratory operations. For labs that fall under the jurisdiction of FDA compliance, 21 CFR Part 11 specifies requirements for electronic data collection is trustworthy, reliable, and equivalent to paper records. FDA regulated labs require solutions that offer complete audit trails. Events such as user logins and changes to key parameters that may affect process outcomes must be tracked. The lab monitoring system must also include user management and data security features such that only authorized users may access the system, that data measurements are non-editable, and that event details are time-stamped to ensure data integrity.
- IMPROVE YOUR DATA INTEGRITY Lack of repeatability and reproducibility of results is a common problem
 for many researchers. The inability to confidently repeat protocols and achieve identical outcomes results in enormous amounts of wasted time for scientists and delays in research progress. Much of the problem surrounding irreproducibility of data lies in a lack of information and insights into how ambient conditions can influence experiments. Fortunately, advanced laboratory monitoring systems are a simple remedy to the problem. By reviewing data for equipment status and ambient conditions, researchers are able to rapidly identify issues contributing to experimental variability and rule out those factors that are not actually affecting experimental results. Furthermore, researchers are able to refine protocols by specifying optimized environmental and instrument parameters to achieve the desired results.
for many researchers. The inability to confidently repeat protocols and achieve identical outcomes results in enormous amounts of wasted time for scientists and delays in research progress. Much of the problem surrounding irreproducibility of data lies in a lack of information and insights into how ambient conditions can influence experiments. Fortunately, advanced laboratory monitoring systems are a simple remedy to the problem. By reviewing data for equipment status and ambient conditions, researchers are able to rapidly identify issues contributing to experimental variability and rule out those factors that are not actually affecting experimental results. Furthermore, researchers are able to refine protocols by specifying optimized environmental and instrument parameters to achieve the desired results.
- MANAGE RISK AND SAVE MONEY Cost savings are often at the core of managing any laboratory budget.
 By utilizing a laboratory monitoring system, lab managers can avoid the time-consuming expense of facility walk-throughs and paper-based recording processes. Automated email and/or SMS alerts of instrument failures can prevent loss of valuable, and often irreplaceable, laboratory materials and specimens. Finally, equipment utilization data can offer insights on equipment usage patterns and influence purchasing decisions regarding capital expenditures.
By utilizing a laboratory monitoring system, lab managers can avoid the time-consuming expense of facility walk-throughs and paper-based recording processes. Automated email and/or SMS alerts of instrument failures can prevent loss of valuable, and often irreplaceable, laboratory materials and specimens. Finally, equipment utilization data can offer insights on equipment usage patterns and influence purchasing decisions regarding capital expenditures.

